SHENZHEN PROKAN ELECTRONICS INC.
Is a high-tech company, which providing integrated solution through development, manufacturer, sales and after-sales service. As one of the most effective medical equipments supplier in global market, Prokan insists on people-for most and market-oriented, uphold creativity and care customers, and always devoting himself to research and manufacture the clinical medical equipment. We are the reliable partner (approved ISO9001,13485) for ODM with the newest technological innovation and patent .Prokan has obtained spectacular achievements in developing and applications of medical testing instruments.
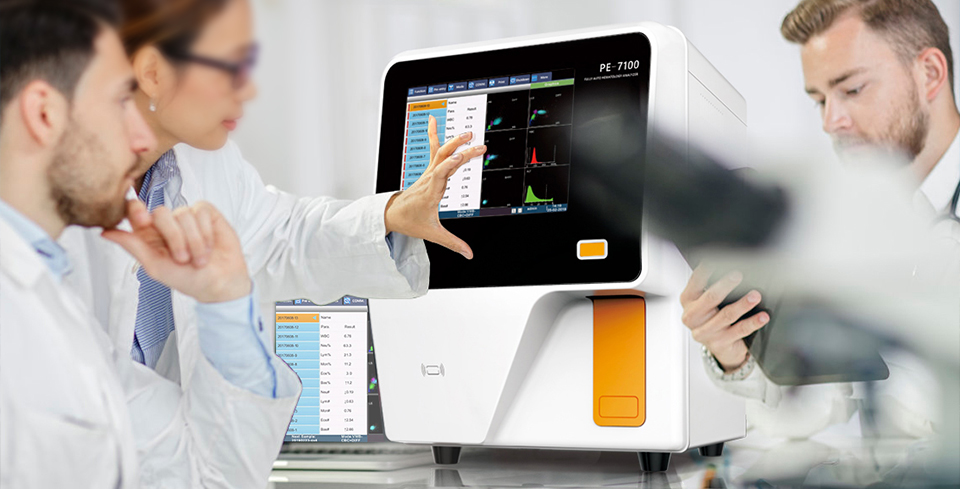
Prokan’s price-performance products are being extended all over the world, including hematology analyzers, biochemistry analyzers, and relevant reagents. Our products which adopt the first-class and advanced international technology of clinical laboratory are successfully sold in domestic and overseas market. It has achieved extensive compliment and approved by our clients in global market. With the broad-ranged application, perfect quality, simple operation, and reliable performance, our products can satisfy your requirements, not only in its quality, but also in its safety standard.
We have a clannish, responsible, professional and efficient team. It is a team based on the development of the company. This team massed the excellent people of different failed, we have rich experience management team and production team, high graduated technicians, high trained professional service-people, to supply continuous service for our customers, under the invariable policy of “High Efficiency, Best Satisfaction”.